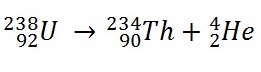In fact the famous E=mc2 equation states that any mass is associated with energy and vice versa; we can’t talk about conservation of energy or conservation of mass alone.
Let’s consider the equation,
Here the reactant loose 0.00459 gm mass, because this amount of mass is converted into energy. This is how a nuclear bomb may destroy a city. So we need to find something which looses significant amount of mass while decaying (fission reaction) or while making larger nucleus (fusion reaction). Uranium and hydrogen are that kind of thing.
So in physics it is clear that mass or energy alone never obey law of conservation.
What happens in chemical reaction? Is mass energy relation applicable in chemistry?
Perhaps you would have read that chemical reaction follows conservation of mass law. Question is how the heat or light comes from a chemical reaction?
In fact mass-energy is also applicable for chemistry. Mass is gained or loosed in any chemical reaction where energy is absolved or evolved.
Let’s consider the equation,
C (graphite) + O2 (g) → CO2; ΔH=-393.5 KJ
So the system looses 393.5 KJ energy and this energy comes from the lost mass, here 4.37×10-12 kg mass is lost. But problem is it’s too small to measure, even with the most sophisticated balance.
Mass of a particle or body in motion
The rest mass is always small. When something is in motion it has kinetic energy. To fulfill E=mc2 condition it gains mass, the greater the speed, the greater the mass.
So question is, the mass or the energy comes from where?
Newton’s 1st law says, ‘Every body persists in its state of rest or of uniform motion in a straight line unless it is compelled to change that state by force impressed on it.’
That means we do not need to consume energy to maintain the speed unless there is any friction or other opposing force. Compared to a particle at rest, a particle in motion has kinetic energy. This energy comes from the force initially applied to make the motion. That means a football may gain some tiny amount of mass when it is kicked!